Mater is made of atoms; such is the absolute truth that governs every single branch in chemistry. Chemists consider atoms as the smallest unit that defines elements along with all their isotopes; they are responsible for all matter liquid, solid or gaseous. To understand why mater behaves the way it does, chemists try to make atomic models. These models are often misunderstood as descriptions of reality but they are, in fact, a representation of what chemists believe reality would look like in order to explain certain phenomena.
In this project, I explored with the imperfection of scientific models by taking inspiration from a well-known anecdote in Antoine de Saint-Exupery’s “Le Petit Prince”. In the book, the protagonist is repeatedly asked by a boy to draw him a lamb. Each time the protagonist draws a lamb, however, the boy is never satisfied with the result. In the end, the protagonist gives up and draws a box in which he says that there is a lamb. For my story, I revisited this concept in the form of the search of an atomic model that explains everything. The same boy from Saint-Exupery’s book is looking for a model of the atom, which explains why the entropy in his asteroid is increasing. He will visit the asteroids of some of the greatest minds of the 20th century searching for answers. Each asteroid will teach the boy and the reader something about an atomic model and some fundamental big ideas like entropy or periodicity, for instance.
For this project, I tried subdividing each concept that I wanted to convey through sketches of the asteroids that I mentioned earlier, along with some dialogue to tell the story as the concepts unfold. Each asteroid’s personality and look is supposed to either represent the nature of the atomic model in question or have a hidden meaning that is explained by the model.
The first asteroid is the boy’s (aka The Little Prince) home asteroid and introduces us to the concept of entropy. It is spiraling towards total chaos due to increasing volcanic eruptions on its surface. Entropy is chaos in its purest form or it is sometimes referred as “the absence of information”. In the universe, entropy tends to increase, as dictated by the second law of thermodynamics.
Upon realizing this, the boy goes and asks Jackson Pollock how to stop entropy from increasing. The painter’s asteroid, constantly being covered in paint, represents the second property of entropy. Entropy tends to increase, unless energy is added to prevent the system from loosing information and spiraling towards complete chaos. The paint represents the energy being added to the asteroid (or system) to preserve the information on its surface. If Pollock were to stop throwing paint at his asteroid, the paint would eventually fade away and make the asteroid loose information. The boy now understands that he can’t stop entropy from increasing, but he can, however, add energy to his asteroid to keep the volcanoes at bay.
He then visits Dalton’s asteroid that is in the shape of a giant grandfather clock whose pendulum is eternally going from one side to another. The Little Prince asks Dalton, the creator of the first scientific model of the atom, how to make energy. Dalton explains energy conservation, the universal law in which energy can neither be created nor destroyed. The pendulum acts as a real-world example of energy conservation since the pendulum’s energy goes from potential to kinetic, forever.
Since energy cannot be created nor destroyed, Dalton gives The Little Prince his model of the atom that looks like a marble to explain entropy. He doesn’t like it since it doesn’t explain any of the properties of matter except gas motion, so he leaves to another asteroid.
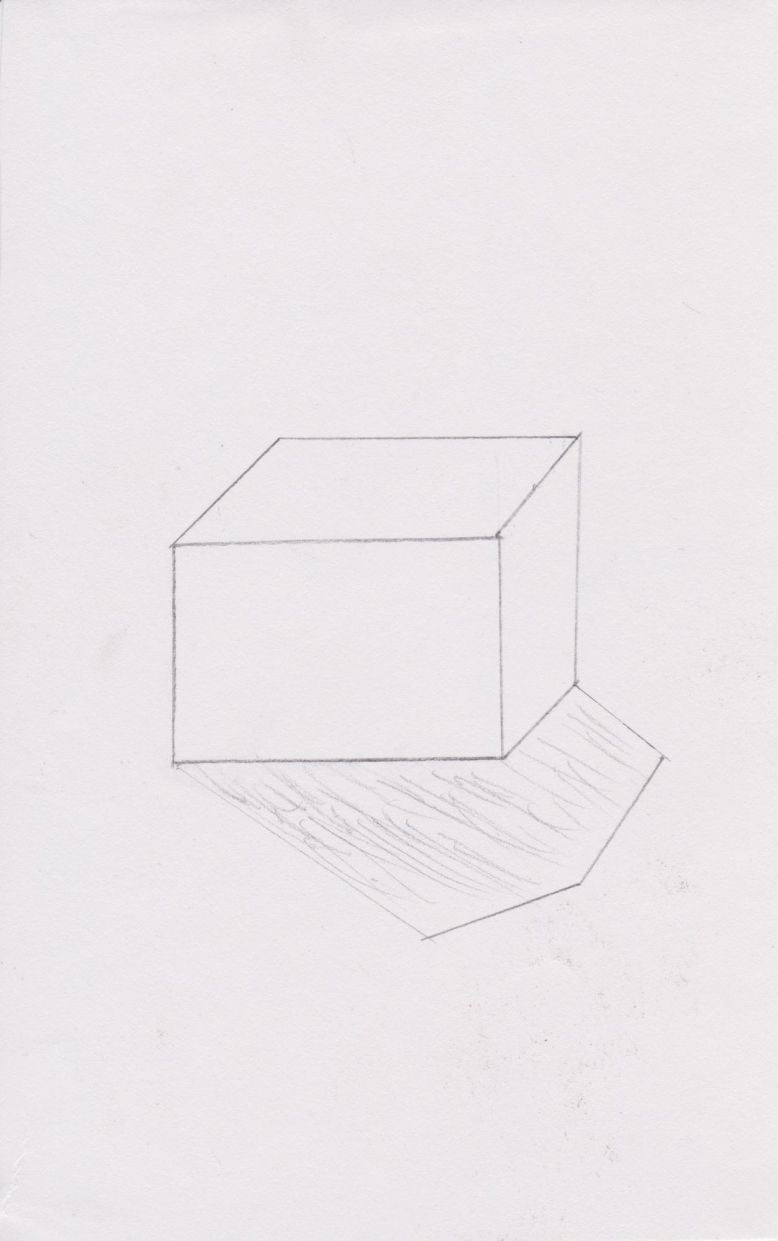
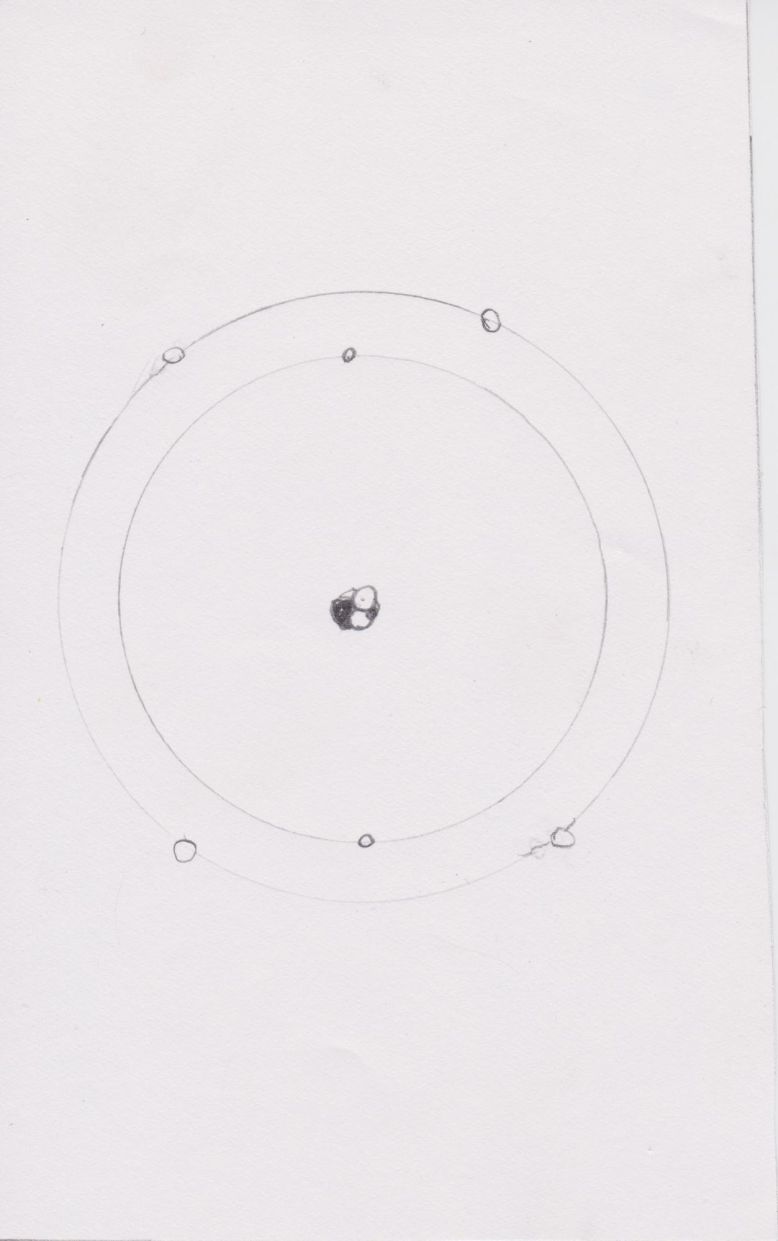
The boy now travels to Rutherford and Bohr’s asteroid, which looks like a cube inside a sphere (as way to try to reconcile the two aspects of their theories). Their model partly obeys Newtonian physics and makes new suppositions that obey quantum mechanics. The electrons are moving around the atom like planets around a solar system, but they also disappear from one energy level and reappear in another. In Bohr and Rutherford’s model, electrons are said to be quantized because they do not follow conventional trajectories, they simply disappear from point A and reappear in point B. Bohr and Rutherford’s asteroid follows these properties; it has both a sphere whose tangent lines are slowly changing from one to another and a cube that has no transition between tangents, the tangents of the cube dramatically change, like the position of an electron. Bohr and Rutherford show the protagonist their model and the Little Prince complains that it is unstable, so he moves to another asteroid.
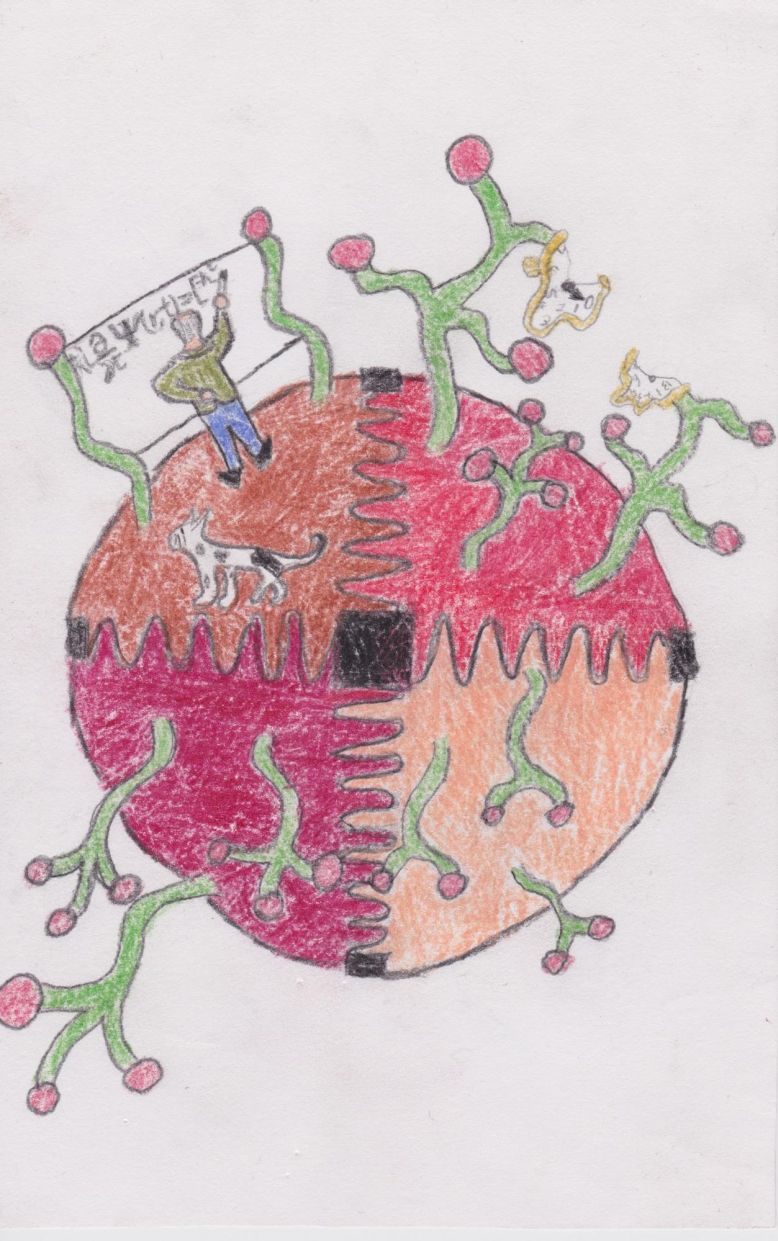
He meets Schrödinger, responsible for the atomic model we use in chemistry today. His asteroid is a big sphere cut into four sections by a sinusoidal wave functions. This is supposed to represent the wave particle-duality properties of his model, a property in which electrons can behave both as a particle (a sphere) or a wave (sinusoidal function). Moreover, all four sections of the asteroid are covered in their respective type of tree, which are wavy and have a ball at its tip. One section’s trees will have the same number of branches, but none of the trees are identical. While I was working on this asteroid, I visualized the periodic table as a forest in which each element is a species of tree. In a forest, there will likely be a general type of tree like the great conifers or the ferns. They share similar properties without being of the same species. I used this as an analogy to explain periodicity; elements of a same group will tend to have similar properties without being the same element. Of course, the trees symbolize the wave and particle properties of mater as well as periodicity. Though it explains many properties, Schrödinger’s model still can’t explain exactly why entropy is increasing.
The Little Prince then leaves Schrödinger’s wave-particle duality wonderland to visit Rothko’s asteroid. Rothko was a cubist painter in the 70's, so his asteroid was a perfect candidate to represent order as a conclusion of my story. Rothko tells the boy that he does not know how to solve the entropy problem, but he can, however, give him a model of the atom that answers all of his questions. He draws him the infamous box from Saint-Exupery’s story that I mentioned at the beginning. The little prince is happy with this drawing since a model is always an artistic representation of a general idea that we use to explain phenomena. The box contains all the hidden knowledge that science has yet to crack open and also all the beauty of the universe.