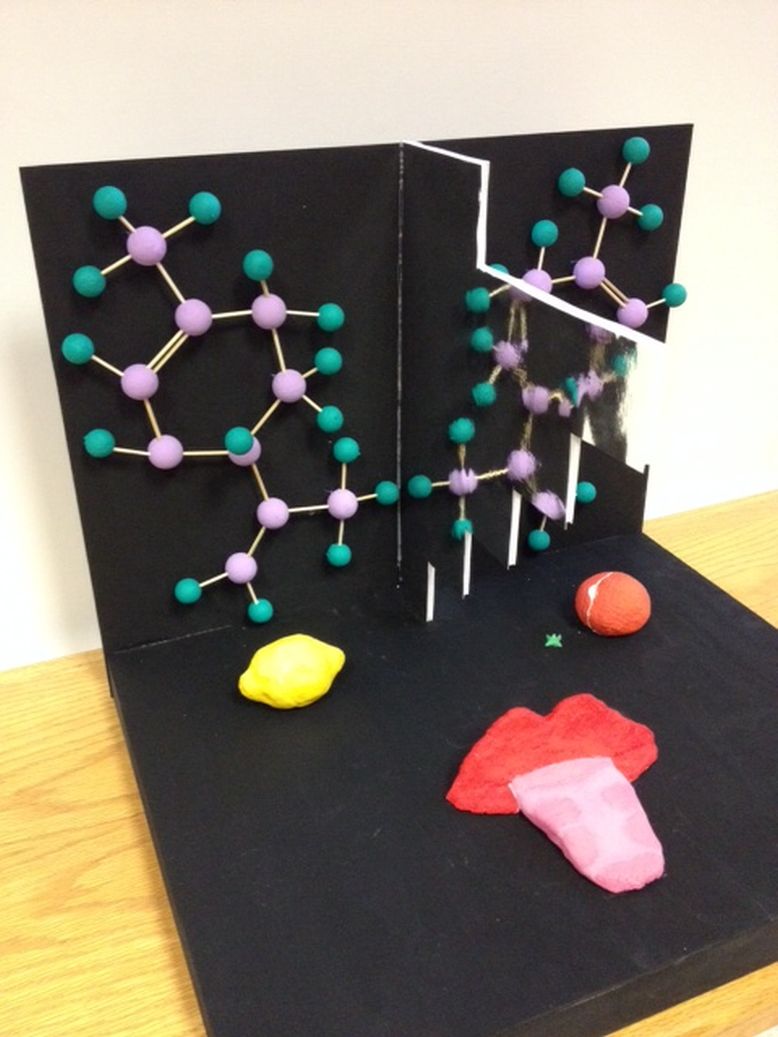
Individuals love the taste of fresh oranges and lemons; little do they know they are essentially the same thing. Molecules may have many different characteristics based on the rotation of their chiral centers. Limonene, a chemical found in citrus fruit peels, has the ability to taste and smell like either oranges or lemons depending on its rotation. It interacts with the nervous system through sensorial receptors on the tongue, allowing us to experience different tastes. Limonene has two molecular structures that are enantiomers, meaning that they contain the same amount of carbon and hydrogen atoms, yet they are non-superposable mirror images of each other. In effect, each one of these structures creates its own product, in this situation being whether it tastes like lemon or orange.
My artwork consisted of displaying both tastes of limonene when it comes in contact with the taste receptors on the tongue. Through my artwork, I was able to display five of Atkins’s big ideas in Chemistry. The simplest and most basic idea is that matter is made up of atoms. This can be clearly seen in my artwork by the two molecular structures of Limonene. Since Limonene has a molecular structure of C10H16, I was capable of clearly representing how the molecule was made up of atoms. The two structures also display how chemical bonds form when electrons pair. In Limonene, all ten of the carbon atoms follow the octet rule, where eight electrons are found on the valence shell. Another one of Atkins’s big ideas is displaying how molecular shape is a crucial feature in chemistry. Although both molecules are Limonene, when the structure becomes a mirror opposite, there is an effect on the sensory receptors, changing the flavour between orange and lemon. Therefore, the slight changes in the structure of the molecule have a large impact on its characteristics, which were displayed by a mirror that was placed down the middle of my artwork. The two Limonene structures are non-superposable, meaning that one cannot be placed on top of another in hopes of seeing them as identical. These three of Atkins’s big ideas are well portrayed through the two molecular structures of Limonene that I created in my artwork.
Two more of Atkins’s big ideas are visible in my artwork through the use of extra items. Since Limonene’s molecular formula is C10H16, we are able to see that both elements are non-metals, therefore allowing me to display periodicity. It is seen in my artwork by the use of a mirror shaped as an upside down staircase. This mirror shape was used to represent the non-metal section of the periodic table. Atkins’s big idea in chemistry that there are residual forces between molecules is displayed through my use of an orange and lemon. Limonene is found in the peels of both of these citrus fruits, yet there are many molecules of Limonene located there. My artwork indirectly portrays how there are dipole-dipole forces between all of the limonene molecules, which result in the creation of condensed matter. Whereas three of Atkins’s big ideas in chemistry where portrayed through the use of Limonene’s structure, another two were displayed through the fruits and a specifically shaped mirror.
A sculpted mouth was placed right in the center of my artwork. On the tongue, the sensorial receptors were added in a darker pink tone in order to display the interaction between the Limonene and the taste buds. The four original tastes (sour, salty, sweet and bitter) are located on the tongue. When Limonene interacts with the sensorial receptors on the tongue, a reaction takes place, forming the distinct taste of either lemon or orange. When Limonene’s stereochemistry is R, it produces the taste and smell of oranges; whereas when the rotation is S, it produces the taste and smell of lemons.
Through my artwork, I tried to display the importance of the rotation of chiral centers. A molecules change in rotation can cause it to have different characteristics and causes it to become an enantiomer. Through the use of the molecule Limonene, I was clearly able to display how matter is made up of atoms, how elements display periodicity, how chemical bonds form when electrons pair, how molecular shape is a crucial feature in chemistry and finally how there are residual forces between molecules.
Sources
RSC - Skeletal chemistry." Web. 28 Nov. 2013.