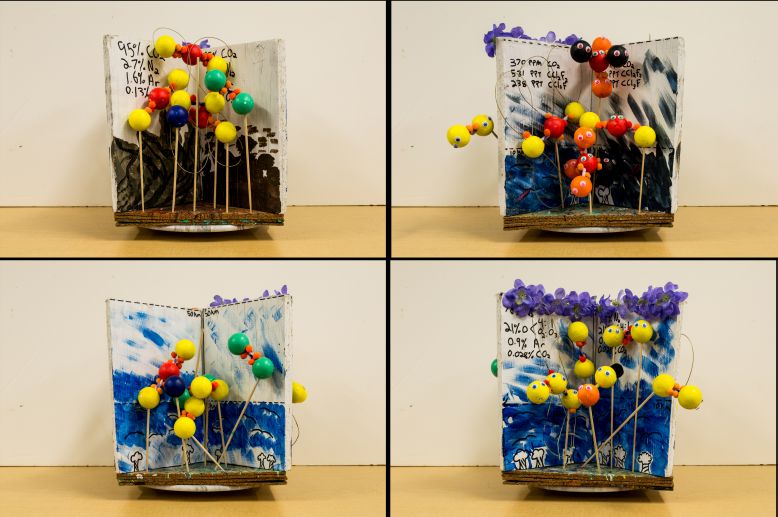The earth's ozone layer is one of the most essential requirements to maintain life on earth. The ozone layer is not actually a layer but it referred to in this way because of the high concentration of O3 molecules in this region with respect to its concentration closer to the surface of the earth. The ozone layer is located at an altitude between 10 km and 50 km. The high concentration of ozone in this region, referred to as the stratosphere, acts in equilibrium with many other molecules such as O2 and CℓO. This equilibrium state allows for ultraviolet radiation to be absorbed. This protects life on earth from the high-energy electromagnetic radiation. Without an ozone layer in the earth’s atmosphere, ultraviolet radiation emitted by the sun would bombard the entire surface of the earth and life would not be able to flourish. This would make for a planetary surface much closer to that of mars in which the rocky terrain cannot support life. Unfortunately, because of the actions of human being, the molecular composition of the ozone layer is changing for the worse. There are many pollutants being emitted such as CCℓ3F and CCℓ2F2, as well as dangerous amounts of CO2. These molecules raise the concentration of chlorine atoms in the ozone layer, which react with lone oxygen atoms. This distorts the equilibrium of the ozone layer and allows for some ultraviolet radiation to pass through our atmosphere and onto the surface of the earth.
My art project represented these various stages of atmospheric equilibrium and distorted equilibrium as well as the reasons for the state of the surface of mars. One way I did this was by showing how the surface of earth differs from that of mars. I painted three quarters of my base using colors such as blue and green to represent life existing on the surface of earth. The other quarter of my base was painted colors such as black red and orange to represent rocks and the lack of life on the surface of mars. I constructed molecules that were present in the type of atmosphere I was depicting and placed them on top of the corresponding surfaces. This allowed me to include four of Atkins's big ideas, namely that; matter is made up of atoms, chemical bonds form when electrons pair, molecular shape is a crucial feature in chemistry, and there exists residual forces between molecules. I also displayed the conservation of energy of this process by showing that bond breaking is an endothermic process. The final big idea that I explored was that elements display periodicity. I represented this by making it seem as if the elements involved in this process acted as if they had personalities.
Aside from certain exceptions, molecules form when electrons pair in a way that follows the octet rule. Sometimes, this process involves hybridization. Hybridization is the mixing of the native atomic orbitals to form special orbitals. This process is assumed to be true based of experimental and observational research, as it is the only model that accounts for the way in which molecules are formed. Hybridization is a crucial feature required to explain the molecular shapes and behaviors of molecules that have been experimentally verified. Molecular shape however, is not the only factor that determines the behaviors of molecular bonding, since residual forces between molecules also have an impact. For example, when an O2 molecule is struck by ultraviolet radiation, the bond between the two oxygen atoms is broken which results in a momentary state in which there are lone oxygen atoms. The reason the N2 cannot absorb this radiation is because it's bond energy is 941 kJ/mol, as opposed to 494 kJ/mol for O2, therefore the energy of ultraviolet radiation is not high enough to break a N2 triple bond. After on O2 molecule is struck by ultraviolet rays, an O2 molecule bonds with a lone O atom to make O3. This structure is angular and is also a resonance structure. This means that because of the polarity of the two oxygen atoms at the end of ozone, by VSEPR (valence shell electron pair repulsion), the shape of the molecule must be angular in order to account for the polarity of the molecule. A resonance structure is present in ozone, meaning that there is one pair of electrons that are not localized. This pair of electrons then forms a bond that makes a structure that is equally polar towards either end of the ozone molecule. This however makes for a weaker bond between each bond between each pair of oxygen atoms. When this happens, a Cℓ atom is able to break one of these bonds because of its relatively high electronegativity. This however forms a free radical because of the odd number of electrons present in the molecule CℓO. This occurrence is not necessarily a bad thing as this leaves an O2 molecule that can proceed to absorb more electromagnetic radiation. The CℓO molecule is short-lived as it inevitably passes by a lone oxygen atom that has an electronegativity value even higher than that of chlorine. This results in another O2 molecule, and a free chlorine atom. This is the way that the equilibrium of the ozone layer works. A disruption in this equilibrium occurs when there are too many molecules containing chlorine atoms. These come from the breaking down of molecules such as CCℓ3F and CCℓ2F2 when absorbing ultraviolet radiation. This results in a higher concentration of momentary CℓO molecules, and therefore a lower concentration of O2 molecules. This phenomenon is commonly referred to as ozone depletion. A lower concentration of O2 molecules results in higher than normal levels of ultraviolet radiation being allowed to pass by the earth's atmosphere and onto to surface of the earth. These harmful molecules, sometimes referred to as CFCs, were not present before the industrial revolution. They mainly are used in aerosol cans and as refrigerants. Though these harmful molecules are slowly being phased out in our society, they have a very long life span (up to one hundred years) because of their inertness and lack of solubility in water. The current relative abundance of CCℓ3F and CCℓ2F2 in our atmosphere is 238 ppt and 531 ppt respectively.
Constructing my art project helped me to better understand what was going on, not only in the atmosphere, but also with chemical bonding in general. By artistically representing natural occurrences, I was able to conceptualize Atkins's nine big ideas in chemistry with a visual aid. Also, the physical aspect of the construction of my artwork forced me to have a clear understanding of the actions and reactions occurring around me. These representations also helped me to understand and appreciate the importance of a model in chemistry in order to comprehend and explain how and why things act the way they do. The best part of these understandings is that the medium used to depict ozone's function, importance, and potential threats, required artistic expression.
Works Cited
http://www.pollutionissues.com/Br-Co/CFCs-Chlorofluorocarbons.html#b
http://www.cem.msu.edu/~reusch/OrgPage/bndenrgy.htm
Yellow Chemistry NYA information sheet provided by vainer college.
Zumdahl Chemistry 9th edition textbook.